
Jozsef Kupai
Budapest University of Technology and Economics, Hungary
Title: Synthesis and application of β-cyclodextrin-based cinchona organocatalysts and their recovery by organic solvent nanofiltration
Biography
Biography: Jozsef Kupai
Abstract
Organocatalysis, in which organic molecules catalyze single or multiple chemical transformations, has emerged as an efficient solution for the rapid and stereoselective synthesis of enantiomerically enriched molecules. Due to the many advantages of organocatalysis compared to conventional metal catalysis, organocatalytic methodologies have become an attractive synthetic tool in asymmetric catalysis.
Cinchona alkaloids and their derivatives have proven to be powerful organocatalysts owing to their reactivities, leading to high enantioselectivities. The presence of tunable functional groups enables cinchona alkaloids to catalyze a broad range of chemical reactions. Chiral thioureas and squaramides are promising classes of cinchona-based organocatalysts. Based on non-covalent interactions they behave as very efficient, directional hydrogen-bond donors.
Nowadays more and more attention is paid to protect our environment, and catalyst recovery can be a useful tool to reach this goal. Organic solvent nanofiltration (OSN) is an emerging technology that allows separation of solutes between 50 and 2000 g•mol-1 via pressure gradient in organic media. Attaching cinchona moieties to β-cyclodextrins (see Figure 1.) seems to be an effective way to reach convenient organocatalysts with high MW, leading to easy separation of the catalyst from the reaction mixture.
New β-cyclodextrin-cinchona thiourea and squaramide organocatalysts were prepared starting from commercially available β-cyclodextrin and hydroquinine. These bifunctional organocatalysts were applied successfully (up to 95% yield and 99% e.e.) in asymmetric Michael additions between 1,3-dioxo compounds and β-nitrostyrene.
After the enantioselective reactions, separation of the products and the CD-based catalysts were carried out applying the OSN method with high efficiency resulting in a green and sustainable organocatalytic method.
This work was supported by the Hungarian Scientific Research Fund/National Research, Development and Innovation Office [OTKA 112289, PD108462].
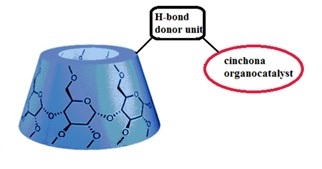
Figure 1. General structure of the new cyclodextrin-cinchona organocatalyst family.

